 No other systemic signs of disease activity were present. Ophthalmologic evaluation revealed severe left eye panuveitis compatible with Behçet uveitis, probably vaccine induced. During the preceding months, the disease was quiescent, being maintained on colchicine therapy, 0.5 mg twice daily.īlood tests showed leukocytosis and elevated CRP and erythrocyte sedimentation rate (ESR). Familial Mediterranean fever (FMF) was previously ruled out, in the absence of typical episodes of fever and abdominal pain. The patient’s medical record was notable for Behçet’s disease, diagnosed four years earlier, manifesting with oral aphthous ulcers, pericarditis, and erythema nodosum. His symptoms began 10 days after receiving the first dose of the vaccine. 
Left eye panuveitis as a manifestation of Behçet’s disease flareĪ 28-year-old male patient (patient #2) presented with a 2-day history of left eye pain, redness, and blurred vision. Upon gradual steroid tapering over a period of 8 weeks, polyarthralgia reappeared, necessitating the commencement of methotrexate therapy.ģ.2. Therapy with prednisone 10 mg brought immediate relief. Autoimmune polyarthritis, possibly representing early rheumatoid arthritis, induced or triggered by the vaccine, was diagnosed. While the acute development of symptoms in proximity to vaccination argue against a diagnosis of rheumatoid arthritis, the distribution pattern and ACPA positivity support the diagnosis. Hand radiographs and chest X-ray were unremarkable. Anti-citrullinated protein antibody (ACPA) was mildly elevated. Antinuclear antibody (ANA), Rheumatoid factor (RF), and parvovirus IgM, tested immediately following the acute symptoms, were negative. Blood tests showed slightly elevated C-reactive protein (CRP). No history of gout, psoriasis, inflammatory bowel disease or recent infection was elicited. Physical examination revealed prominent symmetrical arthritis in the metacarpophalangeal and proximal interphalangeal joints. His medical history was notable for dyslipidemia, non-alcoholic fatty liver disease, and active smoking. His complaints appeared three days after receiving the first dose of the vaccine. 
Here, we present eight cases of autoimmune phenomena following COVID-19 vaccination, suggesting a possible association between these novel vaccines and autoimmunity.Ī 49-year-old male (patient #1) presented with a 2-day history of bilateral hand pain, swelling, and stiffness. This tremendous vaccination rate provides an opportunity to detect rare adverse events not reported in the original trial. Studies suggest significant efficacy in preventing COVID-19 and reducing disease severity. This is especially the case for the mRNA-based vaccines available, the first mRNA-based vaccines entering mass use.īy March 2021, more than half of the adult (>16 years) population in Israel had been vaccinated with at least one dose of the BNT162b2 mRNA coronavirus disease 2019 (COVID-19) vaccine (BioNTech and Pfizer). Although direct causation is debatable, the association is plausible.ĭetecting immunological adverse reactions to the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines is of great public and scientific interest. Many vaccine related immunological adverse events have been described for example, evidence for an increased risk of Guillain-Barre syndrome following Influenza vaccine, an association between systemic lupus erythematosus and the papilloma vaccine, and episodes of immune demyelination after hepatitis B vaccine were all previously suggested. Vaccines as triggers of autoimmunity is a controversial subject. We briefly expound on the theoretical background of vaccine related autoimmunity and explore future research prospects. We observed that while immune phenomena may occur following vaccination, they usually follow a mild course and require modest therapy. All patients were assessed in our tertiary care center in Israel and had no history of previous SARS-COV-2 infection. We report a series of patients presenting with de-novo or flares of existing autoimmune conditions associated with the Pfizer BNT162b2 mRNA SARS-CoV-2 vaccine. In Israel, where a large percentage of the population has been rapidly vaccinated, such adverse events may be more apparent. 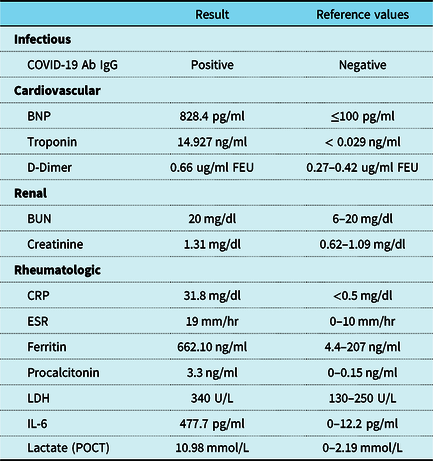
While studies indicate the vaccine is exceptionally safe, rare systemic side effects remain possible. Widespread vaccine distribution has yet to occur in most countries, partially due to public concerns regarding possible side effects. Vaccines represent an attractive possible solution to the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) pandemic. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
